Do you have a GSD news story?
Message us, or see the news section in Notes for contributors.
15th April is International Pompe Day.
Find out more about what a typical day might look like for community members affected by Pompe and what they find most helpful.
McArdle walking courses open
Full details of the 2025 McArdle walking courses are now available, and we are open for bookings.
Rare Disease Day 2025
Today is Rare Disease Day!
Research opportunities for people affected by hepatic GSDs.
Find out about how to get involved in current hepatic GSD research studies.
Update on the Beam GSD1a programme.
Beam Therapeutics have put out a press release that includes an update on U.S. Food and Drug Administration clearance for their investigational new drug application for BEAM-301 in GSDIa.
Virtual AGSD-UK support.
Virtual Groups As children are returning to school, AGSD-UK will be holding a virtual group for parents or carers who might have questions around SEN, anxiety, or behaviour. If you would like to attend on the 9th September at 7.30pm please email info@agsd.org.uk....
Update on Spark Therapeutics Pompe gene therapy programme.
Spark have provided the following community update on their Pompe gene therapy programme.
Update on phase 3 study of DTX401.
Ultragenyx have provided the following community update on the phase 3 study of DTX401, their investigational gene therapy for the potential treatment of GSDIa. GSD1a Community Update
International Pompe Day.
Find out about new Pompe information resources
Save the Date!
Main Event 2024
Rare Disease Day 2024.
Today is Rare Disease Day! This rarest of days, 29th February is a great opportunity to raise awareness about rare conditions. Visit https://www.rarediseaseday.org/ to find out more about how to get involved in this international campaign. Thanks go to all our...
Cipaglucosidase alfa (Pombiliti®) is accepted for use within NHS Scotland.
Cipaglucosidase alfa (Pombiliti®) is accepted for use within NHS Scotland The Scottish Medicines Consortium has completed its assessment of cipaglucosidase alfa and has advised that this offers an additional treatment choice as a long-term enzyme replacement therapy...
Virtual Festive Event.
AGSD-UK is holding a Virtual Festive Event on the 9th of December at 1.00pm. Come along and make sure you wear a Festive hat, as there will be a prize for the best headgear. We will have a Quiz and time for everyone to catch up. For more information, please contact...
Update on the DTX401 programme for GSD1a.
Ultragenyx have provided the following community update on the development of DTX401, their investigational gene therapy for the potential treatment of GSDIa. GSDIa Community Update
Newborn Genomes Programme.
GSDs on list of conditions for inclusion in Newborn Genomes Programme research study. Genomics England has published an initial list of conditions that will be screened for as part of their forthcoming Generation Study, which includes some GSDs. The study will...
NICE recommends cipaglucosidase alfa plus miglustat.
The National Institute for Health and Care Excellence has today published final guidance recommending cipaglucosidase alfa (CIPA) plus miglustat, within its marketing authorisation, as an option for treating late-onset Pompe disease in adults.
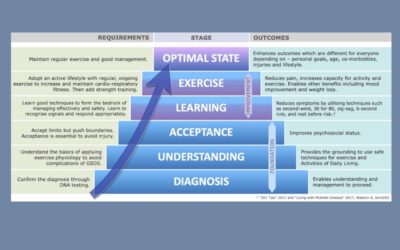
New Continuum of Care Model for McArdle’s.
IamGSD has led an international team of clinicians to develop and publish a continuum of care model to help physicians and patients to achieve the patient’s optimal state.
Seminar for people affected by neuromuscular diseases.
Seminar Thursday 18th May - 5PM UK The research team at Newcastle University's John Walton centre are keen for people affected by neuromuscular conditions to join a seminar on 18th May: ‘We are glad to announce that we will be delivering a seminar on the 18 May at...
Main Event 2023 Programme
Check here for latest programme details and to register for the event.
International Pompe Day 15 April 2023.
The IPA focused this year’s International Pompe Day on the importance of movement, sharing stories from the community under the slogan “Every Move Counts”.
London Marathon 2023.
London Marathon 2023 AGSD-UK has 4 brilliant and brave supporters running the London Marathon on 23rd April. To read their motivation for running please have a look at the individual stories on their fundraising pages. Any donations would be very much appreciated....
Rare Disease Day 2023.
Rare Disease Day is taking place on 28th February 2023 This Rare Disease Day is an opportunity to highlight the importance of effective, coordinated care for people living with rare conditions. Visit the Rare Disease Day 2023 website to find out more! Rare...
Help Us Raise Awareness.
Help us raise awareness and share experiences With rare disease day approaching at the end of February can you help us raise awareness of GSD and what it's like to live with the condition? Just send a short video clip of the way your condition affects a typical...
Newsflash – The Main Event 2023 date change!
Date change for The Main Event 2023
The Main Event 2023.
Register your interest now for The Main Event 2023.
Liberating Research GSD1a Study.
GSD1a Patient Research Please find below a link to an invitation for UK families affected by GSD1a to take part research by Liberating Research to improve understanding of the impact of the condition. Who can participate?People living with GSD1a and their partners /...
McArdle’s walking courses 2023.
The dates for the McArdle’s walking courses for 2023 have been announced. Courses will be in Snowdonia, based in the village of Tremadog.
Great North Run 2022.
This Sunday is the GNR and AGSD-UK have several runners taking part.
LSD Collaborative Community Survey 2022.
Clinical and Social Care Services Please take the chance to respond to an important survey to improve understanding about the quality of care you experience from your specialist hospital clinic, homecare, and other social care services. The survey is designed by the...
International GSD conference 2022.
The International GSD Conference (IGSD2022) will be virtual this year.
NICE recommends Avalglucosidase alfa as a treatment option for Pompe.
24th August 2022 The National Institute for Health and Care Excellence today published its recommendation of Avalglucosidase alfa (AVAL) as an option for treating Pompe in babies, children, young people and adults, where AVAL is provided according to the commercial...
Wales Rare Diseases Action Plan.
Action Plan The Welsh Government have now published their Rare Diseases Action Plan in support of the 2021 UK Rare Disease Framework. The Wales Rare Diseases Action Plan 2022-2026 is available at the following links: English version Welsh version
New and Improved CureGSD1b Website.
The updated CureGSD1b website is now live.
GSD & ME website is updated.
The updated GSD & Me website is now live.
Update on NICE assessment of new treatments.
An update on the appraisal of treatments for Pompe by NICE.
Moderna mRNA trial infusion for GSD1a.
First patient receives Moderna mRNA trial infusion for GSD1a.
Volunteering opportunity at McArdle Clinic.
The commitment is at UCLH, 10am to 4pm every Wednesday, to support the McArdle team and to directly assist and guide patients.
McArdle’s Walking in Wiltshire weekend.
Just a month to go to the “Walking in Wiltshire” get-together weekend for all those with McArdle’s. 17-19 June.
AGSD-UK Help and Support.
AGSD-UK can provide support to individuals and families affected by GSD.
“The McArdle’s Experience” walking course.
The walking course for this year is to be held in the Pembrokeshire Coast National Park, 30 July to 6 August. Full details now available here on our website.